 Consequently, internal friction and viscosity increase. In most liquids, pressure reduces the free volume in the internal structure, and thus limits the movability of molecules. For example, lubricants in cogwheels or gears can be submitted to pressures of 1 GPa and higher.Įquation 4. Highly viscous mineral oils react with a viscosity increase of times 20000 under identical circumstances.įor synthetic oil this pressure change can even result in a viscosity increase by a factor of up to 8 million. This applies to most low-molecular liquids. In case the pressure goes up from 0.1 MPa to 200 MPa, the viscosity can rise to 3 to 7 times the original value. 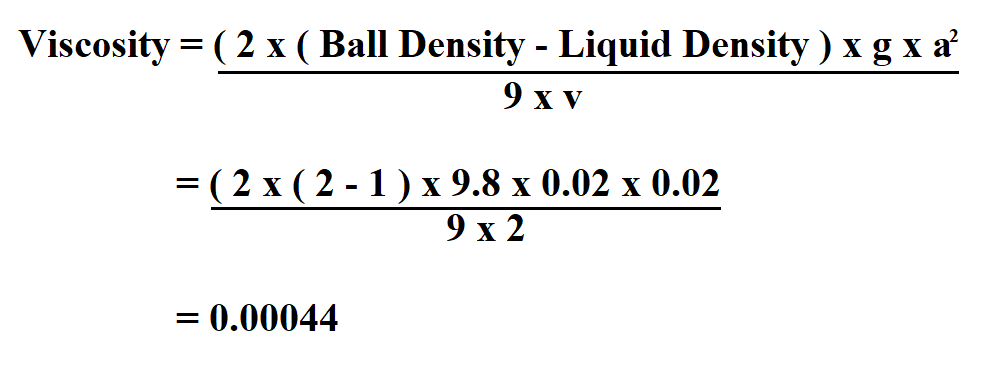 
For comparison: This same change in viscosity is usually provoked by a minor temperature change of 1 K (1 ☌). Most liquids react to a significantly altered pressure (from 0.1 MPa to 30 MPa) with a viscosity change of about 10 %. However, fluids are not dramatically affected if the applied pressure is low or medium: liquids are almost non-compressible in this pressure range. Normally, an increase in pressure causes a fluid’s viscosity to increase, too. Three factors determine a substance’s flow behavior. Still, shear rate is not the only influencer. To learn more consult “ The Rheology Handbook”. The flow behavior of non-Newtonian liquids can be far more complex than these basic examples. yogurt), while for others the viscosity increases with increasing shear rate (curve 3 | e.g. 
If its viscosity changes with the shear rate, a liquid is non-Newtonian and – for exact definition – one has to specify the apparent viscosity.ĭifferent shear-dependent fluids behave differently: For some, their viscosity decreases when the shear rate increases (curve 2 | e.g. Typical Newtonian liquids are water or salad oil. For a Newtonian liquid, this function is a straight line (curve 1) see figure 6. Such fluids are named ‘Newtonian liquids’ after Sir Isaac Newton.Ī viscosity function shows the viscosity over the shear rate. the shear rate – acting upon the fluid, it is ideally viscous. If a fluid’s internal flow resistance is independent of the external force – i.e. High-viscosity fluids resist deformation. Table of content 1 Dynamic Viscosity Formula 1.1 What is Dynamic Viscosity 1.2 The formula for Dynamic Viscosity 2 Solved Examples Dynamic Viscosity Formula What is Dynamic Viscosity It is not easy to find a lot of information on the differences between dynamic and kinematic viscosity. Fluids which flow easily show a low resistance to deformation. viscous fluids that contain an elastic portion. The specific field of viscometry covers ideally viscous fluids, and – considering certain restrictions – also viscoelastic liquids, i.e. sweet jelly) or as a viscoelastic liquid (like e.g. According to a material’s properties, we either classify it as a viscoelastic solid (like e.g. That is, substances which are neither completely elastic, nor entirely viscous. In everyday life, we mostly come across viscoelastic materials. Scientists specify solid materials as being elastic and liquids as being viscous. Imagine all materials as classified on a virtual scale from solid to liquid. Rheology deals with the flow behavior and deformation of materials. Gives the apparent viscosity.In order to determine a fluid’s viscosity, you have to enter the field of viscometry, a subject area of a wider science called rheology. In fluid mechanics, apparent viscosity (sometimes denoted η) is the shear stress applied to a fluid divided by the shear rate: The apparent viscosity of a dilatant fluid is higher when measured at a higher shear rate ( η 4 is higher than η 3), while the apparent viscosity of a Bingham plastic is lower ( η 2 is lower than η 1). The apparent viscosity of a fluid depends on the shear rate at which it is measured. 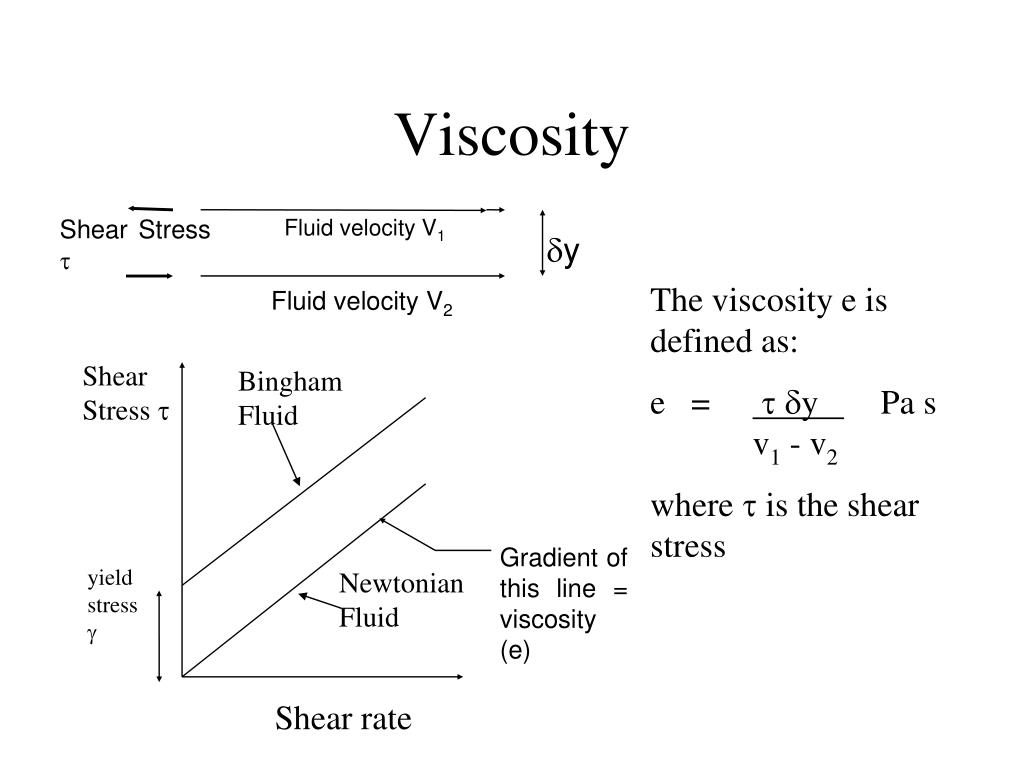
JSTOR ( March 2009) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed.įind sources: "Apparent viscosity" – news Viscosity is the property of fluid which defines the. Please help improve this article by adding citations to reliable sources. For gases we take air or O2 as a standard fluid with density, 1.293 kg/m3. This article needs additional citations for verification. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
